The problem of substandard and falsified (SF) medical products affects all countries but few regulatory authorities or pharmaceutical companies have policies of publicly releasing data. In order to fill the current and emerging evidence gaps, and to improve understanidng of the scale of the global SF medical product issue, the Infectious Disease Data Observatory's (IDDO) Medicine Quality Research Group has developed a system for collating and curating global data on the quality of essential medical products and occurrence of substandard and falsified (SF) medical products in the lay press - the Medicines Quality Monitoring (MQM) Globe.
About the Medicine Quality Monitoring Globe
The MQM Globe holds data from July 2018 and tracks:
- The quality of medicines/medical products, including vaccines, medical devices, traditional and herbal medicines, vitamins, weight loss medicines, nutritional supplement, veterinary medicines, blood products and psychoactive substances, if the quality of these products is suspected.
- Recalls, seizures, diversions, thefts, degradation, adulteration or contamination of medicines, cases of patients suffering adverse effects/lack of efficacy after taking a medicine suspected to be substandard or falsified.
- General discussion, development/marketing of a new system or technologies to identify or prevent poor quality medicines, and articles on new laws/regulations.
The MQM Globe can provide users with customised summaries of national and international newspaper reports on medicines and medical products’ quality, viewable on-screen in French, Spanish, Mandarin and English, and available to download in three languages – English, French and Spanish.
The MQM Globe is free to use and will be useful to researchers, clinicians and policy makers looking to track SF medical products both nationally and internationally.
The MQM Globe and COVID-19
With the recent emergence of COVID-19, there has been a growing number of reports of falsified masks, diagnostic tests, medicines and vaccines, and in light of this, there has never been a more urgent need for a shared global tool to track SF medical products.
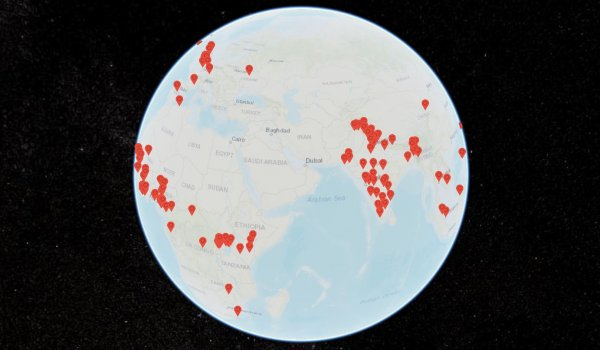
The MQM Globe maps real-time media reports on the quality of medical products across the world. Its database is continually updated and includes medicines, vaccines, biologics and diagnostics and now has a COVID-19 search function to include reports of COVID-19 related SF medical products, including masks, diagnostic tests, medicines and vaccines.
Professor Paul Newton, who leads the Research Group, said: “The Medicine Quality Monitoring Globe will prove a valuable resource to the many individuals, researchers and public health organisations working in this area. For the first time, users can freely access global reports of substandard and falsified medical products in one place to track what may be happening in their communities, their country and around the world. There are a growing number of reports of falsified masks, diagnostic tests, medicines and vaccines for COVID-19 infection and we fear that these will continue to increase.”
Dr Céline Caillet, Group Coordinator commented: “Assessing the scale of the problem globally with new tools like the Medicine Quality Monitoring Globe is a key first step. Little research has been done in this area and there is an urgent need for wider, multidisciplinary research to build the evidence base globally for informing interventions and policy to ensure that we all have access to good quality medical products.”
Please Sign in (or Register) to view further.